Toxoplasma membrane inositol phospholipid binding protein TgREMIND is essential for secretory organelle function and host infection
Cell Rport Volume 43, Issue 1, 23 January 2024, 113601
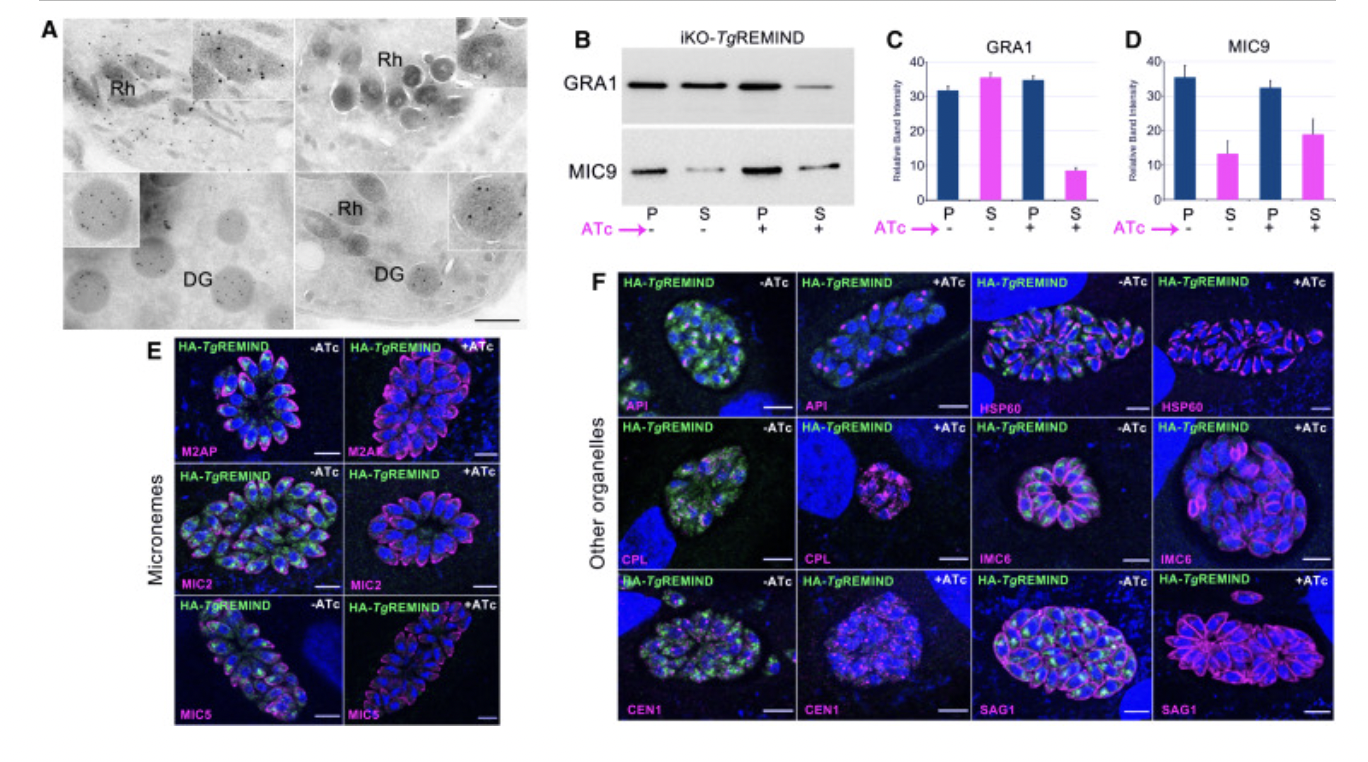
(A) Immunoelectron microscopy using untreated (left) or ATc-treated (right) iKO-TgREMIND mutants followed by staining with anti-ROP1 (top) or anti-GRA1 (bottom) antibodies and revealed by immunogold secondary antibodies. Insets in the top right corners correspond to a magnification of one rhoptry or dense granule. Scale bar, 500 nm.
(A) 使用未经处理的(左)或 ATc 处理的(右)iKO-TgREMIND 突变体进行免疫电子显微镜检查,然后用抗 ROP1(上)或抗 GRA1(下)抗体染色,并用免疫金二抗显示。 右上角的插图对应于一个菱形或致密颗粒的放大倍数。 比例尺,500 nm。
Electron and immunoelectron microscopy
Intracellular parasites were fixed at room temperature for 1h in 0.1 M sodium cacodylate buffer containing 2.5% glutaraldehyde. Then, samples were kept overnight at 4°C. After washing with the same buffer, samples were fixed again in aqueous 1% osmium tetroxide and 1.5% potassium ferrocyanide. After successive dehydration in ethanol, samples were embedded in Epon (Agar Scientific, AGR1165): 2h in resin mixed with ethanol and 2h with pure resin before polymerization 24h at 60°C. Sections of 70 nm were cut using an ultramicrotome LEICA UC6, collected on grids, and contrasted 15min in 2% aqueous uranyl acetate and 2min in Reynold’s lead citrate. Ultrastructural images were collected using a JEOL1400 transmission electron microscope at 80kV and camera RIO9. For immuno-electron microscopy, the HFF cells infected with iKO-TgREMIND mutants treated with ATc for 36 hours or untreated were fixed overnight at 4°C in 8% paraformaldehyde with 0.1% glutaraldehyde in PBS buffer, thoroughly washed in the same buffer, pelleted in 10% gelatin and infused overnight in sucrose 2.3 M containing 20% polyvinyl pyrrolidone 10000 in phosphate buffer 0.1 M. The pellets were mounted on ultracryomicrotome supports and rapidly frozen in liquid nitrogen. Ultrathin sections of about 90–100 nm were obtained using a UC6 ultramicrotome equipped with an FC6 device (Leica). For immuno-labeling, sections were incubated in blocking medium (0.05 M glycine, 5% fish gelatine in 0.1 M PBS buffer) for 30 min. The grids were incubated with rabbit polyclonal anti-ROP1 or monoclonal anti-GRA1 antibodies for 1.5 hour at 37°C. After washing, sections were incubated at room temperature for 30 min in the corresponding secondary gold conjugates (Aurion, 10nm gold-labeled antibody) diluted in the same buffer. Following a final thorough wash in PBS alone, the grids were fixed in 4% glutaraldehyde for 10 min at room temperature and washed in water. After staining 0.5% uranyl acetate in 1.5% methylcellulose, sections were observed on a JEOL1400 transmission electron microscope at 80 kV accelerating voltage.
电子和免疫电子显微镜
细胞内寄生虫在含有 2.5% 戊二醛的 0.1 M 二甲胂酸钠缓冲液中室温固定 1 小时。 然后,将样品在 4°C 下保存过夜。 用相同的缓冲液洗涤后,将样品再次固定在1%四氧化锇和1.5%亚铁氰化钾水溶液中。 在乙醇中连续脱水后,将样品包埋在 Epon (Agar Scientific, AGR1165) 中:在与乙醇混合的树脂中嵌入 2 小时,在纯树脂中嵌入 2 小时,然后在 60°C 下聚合 24 小时。 使用超薄切片机 LEICA UC6 切割 70 nm 的切片,收集在网格上,并在 2% 醋酸双氧铀水溶液中对比 15 分钟,在雷诺柠檬酸铅中对比 2 分钟。 使用 80kV 的 JEOL1400 透射电子显微镜和 RIO9 相机收集超微结构图像。 对于免疫电子显微镜,将用 ATc 处理 36 小时或未处理的 iKO-TgREMIND 突变体感染的 HFF 细胞在 8% 多聚甲醛和 0.1% 戊二醛的 PBS 缓冲液中于 4°C 固定过夜,在相同缓冲液中彻底洗涤,沉淀 在 10% 明胶中,并在含有 20% 聚乙烯吡咯烷酮 10000 的 0.1 M 磷酸盐缓冲液的蔗糖 2.3 M 中输注过夜。将沉淀安装在超低温切片机支架上并在液氮中快速冷冻。 使用配备 FC6 装置(徕卡)的 UC6 超薄切片机获得约 90-100 nm 的超薄切片。 对于免疫标记,切片在封闭介质(0.05 M 甘氨酸、0.1 M PBS 缓冲液中的 5% 鱼明胶)中孵育 30 分钟。 将网格与兔多克隆抗 ROP1 或单克隆抗 GRA1 抗体在 37°C 下孵育 1.5 小时。 清洗后,将切片在用相同缓冲液稀释的相应二级金缀合物(Aurion,10nm金标记抗体)中室温孵育30分钟。 最后仅用 PBS 彻底洗涤后,将网格在室温下于 4% 戊二醛中固定 10 分钟并用水洗涤。 用1.5%甲基纤维素中的0.5%乙酸双氧铀染色后,在JEOL1400透射电子显微镜上观察切片。